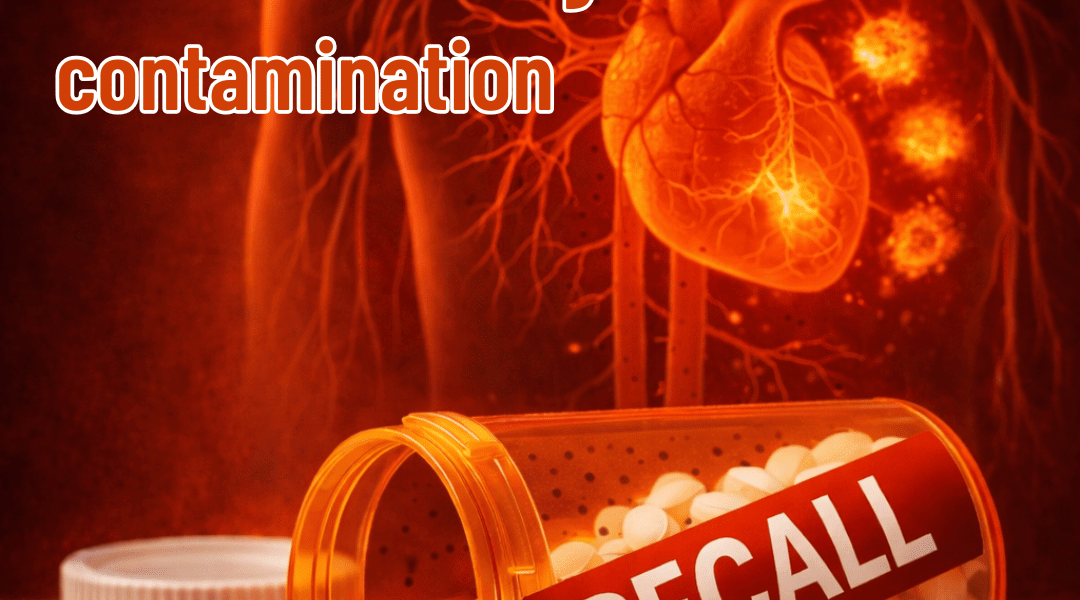
Nearly 600,000 bottles of a common BP drug were pulled from the market due to cancer-causing contamination. What patients need to know
Discovering a recall on a medication you’ve trusted for years to manage your blood pressure can spark immediate worry and uncertainty, especially when it involves a potential health risk like a chemical impurity. That concern grows when headlines mention links to cancer, making you question if your daily capsule is safe or if changes are needed right away. Many people feel this unease, particularly those relying on prazosin for steady blood flow and peace of mind. The reassuring part? Official guidance emphasizes calm action over panic. Drawing from FDA announcements and health reports, this article breaks down the recent prazosin hydrochloride recall clearly—so you can make informed choices with your healthcare provider.

Understanding the Prazosin Recall 🛑
Prazosin hydrochloride is a widely used medication that helps relax blood vessels, supporting healthy blood pressure levels and reducing risks like strokes or heart issues. It’s also prescribed for other concerns, such as sleep disturbances related to PTSD.
Recently, a voluntary nationwide recall affected certain lots of prazosin capsules due to the detection of a nitrosamine impurity called N-nitroso Prazosin impurity C at levels above acceptable limits. Nitrosamines are common environmental compounds, but prolonged exposure to elevated amounts in medications may pose health concerns, including a potential increased cancer risk according to regulatory standards.
This recall highlights ongoing efforts to maintain drug safety, but it’s classified as Class II by the FDA—indicating that any potential effects are likely temporary or reversible, with low probability of serious harm.
Key Details of the Affected Medication
The recall involves prazosin hydrochloride capsules in these strengths:
Distributed by Teva Pharmaceuticals and others, it covers multiple lots with varying bottle sizes (typically 100 to 1,000 capsules each). Not all prazosin is affected—only specific batches tested showed the impurity concern.
Many patients continue their prescriptions without issue from unaffected supplies. The focus remains on quality control in manufacturing.
What Nitrosamine Impurities Mean for Health 🔬
Nitrosamines can form during drug production or storage under certain conditions. While found in trace amounts in foods, water, and air, regulatory agencies like the FDA set strict limits for medications to minimize long-term exposure risks.
Research indicates that high levels over extended periods might contribute to health issues, prompting vigilant testing. This isn’t the first such recall—similar actions have occurred with other medications in recent years, reflecting improved detection methods rather than widespread new dangers.
The good news? Short-term use of affected lots carries remote risk, per FDA classification.

Steps to Take If You’re Taking Prazosin 💊
Feeling unsure about your medication is completely understandable—here’s practical guidance to navigate this smoothly:
- Check your bottle: Look for manufacturer (e.g., Teva) and lot details. Resources from the FDA or your pharmacy can help identify affected batches.
- Do not stop suddenly: Abruptly discontinuing blood pressure medication can lead to rebound effects like elevated pressure. Always continue until advised otherwise.
- Consult your healthcare provider or pharmacist: They can verify if your supply is impacted, discuss alternatives if needed, and monitor your health.
- Return affected products: Follow instructions from your pharmacy or the manufacturer for safe returns.
- Stay informed: Reliable sources like the FDA website provide updates on recalls.
These steps empower you to handle the situation proactively without unnecessary stress.
Comparing This Recall to Broader Trends 📊
Nitrosamine concerns have led to recalls in various drugs over time. Here’s a quick overview:
| Medication Type | Common Impurities Noted | Recall Frequency | Patient Advice |
|---|---|---|---|
| Blood Pressure Meds | Nitrosamines (various) | Occasional | Consult provider; don’t stop abruptly |
| Other Categories | Similar compounds | Past years | Check lots; seek alternatives if needed |
This table shows prazosin joins a monitored group, with emphasis on patient safety through transparency.
But that’s not all—prevention plays a role too.
Tips for Medication Safety Moving Forward
Beyond this recall, simple habits support confidence in your prescriptions:
- Store medications in cool, dry places as directed.
- Review labels and expiration dates regularly.
- Discuss all meds with your doctor during visits, including generics.
- Report any concerns to your pharmacist—they’re a great resource.
- Use official channels for recall alerts, like FDA notifications.
Consistency in these areas often brings the greatest peace of mind.
In Summary
The recall of certain prazosin hydrochloride capsules addresses a nitrosamine impurity concern, but official assessments suggest low immediate risk for most patients. By checking your supply and talking with your healthcare team, you can continue managing blood pressure effectively while staying protected. Knowledge turns worry into empowered action.
Frequently Asked Questions
Is my prazosin affected by this recall?
It depends on the specific lot and manufacturer—check with your pharmacist or official recall lists for confirmation.
Should I stop taking prazosin right away?
No—sudden stops can affect blood pressure control. Always consult your doctor or pharmacist first for personalized guidance.
Are there safe alternatives to prazosin?
Yes, many options exist for blood pressure management; your healthcare provider can recommend the best fit based on your needs.
Disclaimer: This article is for informational purposes only and is not intended as medical advice. It is not a substitute for professional diagnosis or treatment. Always consult your healthcare provider before making changes to your medication regimen, especially with ongoing health conditions.